Research Center for Nerve Reconstruction
R&D > Laboratories > Research Center for Nerve Reconstruction

Our Vision
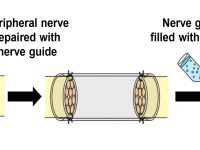
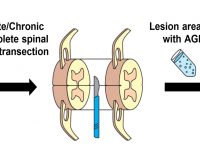
Targeting improvement in nerve reconstruction and rehabilitation from several aspects, aiming at the creation of innovative treatments to both peripheral nerve (PN) and spinal cord (SC) injuries.

Contact Us
Primary Investigators

Prof. Shimon Rochkind, Lab PI
Phone: +972-52-4266442
Email: shimonr@tlvmc.gov.il

Dr. Mara Almog, Lab Manager
Phone: +972-52-5918192
Email: maraa@tlvmc.gov.il
Address
Founders building
5th floor room 21

Research
Gallery




Our Team
Current Staff
- Prof. Shimon Rochkind (MD, PhD)
- Dr. Mara Almog (PhD)
- Dr. Moshe Nissan (PhD)
- Mr. Igal Koifman (MBA)
Past Staff
- Dr. Mira Mandelbaum-Livnat (PhD)
- Prof. Zvi Nevo (PhD) (deceased)
- Dr. Yoram Wollman (PhD)
- Dr. Vadim Poliansky (DVM)
- Mr. Akiva Korn (M.Med.Sc., D-ABNM)
- Dr. Alex Litvak (DVM)
- Dr. Valery Davidson (MD)
Current funding



Highlight Publications
Meyer C, Wrobel S, Raimondo S, Rochkind S, Heimann C, Shahar A, Ziv-Polat O, Geuna S, Grothe C, Haastert-Talini K. Cell Transplant. 2016 Jan 1; 25(1):159-82. |
Peripheral nerve repair and muscle preservation. In: “Handbook of Low Level Laser Therapy”. Rochkind S. Edited by Hamblin MR, Pires de Sousa MV, Agrawal T. |
Photobiomodulation in Peripheral Nerve Injury with Aspect to Muscle Response. Mandelbaum-Livnat MM, Almog M, Nissan M, Loeb E, Rochkind S. Photomed Laser Surg. 2016 Dec 1; 34(12):638-645. |
More Publications >>
Laser Photobiomodulation in Neuroscience: From Bench to Bedside. Rochkind S. Photomed Laser Surg. 2016 Dec 1; 34(12):585-586. |
Rochkind S and Shainberg A. J Reconstr Microsurg. 2017 Feb 20; 33(5):352-357. |
Fregnan F1, Ziv-Polat O, Morano M, Mandelbaum-Livnat MM, Nissan M, Koren A, Biran T, Yifat Bitan, Reider E, Viano N, Almog M, Rochkind S, Geuna S, Shahar A. Edited by Colette Maurício A, IntechOpen. 2017 May 31; chapter 8. |
Photobiomodulation in Neuroscience: A Summary of Personal Experience. Rochkind S. Photomed Laser Surg. 2017 Nov 1; 35(11):604-615. |
Rochkind S, Mandelbaum-Livnat MM, Raimondo S, Morano M, Ronchi G, Viano N, Nissan M, Koren A, Biron T, Bitan Y, Reider E, Almog M, Ziv-Polat O, Shahar A and Geuna S. Edited by Francesco Baino, IntechOpen. 2017 Dec 13; chapter 8. |
Dietzmeyer N, Huang Z, Schüning T, Rochkind S, Almog M, Nevo Z, Lieke T, Kankowski S, Haastert-Talini K. Cell Transplant. 2020 Mar 15; 29:963689720910095. |
Sapir Y, Buzaglo N, Korn A, Constantini S, Roth J, Rochkind S. Childs Nerv Syst. 2021 Jan 6; 37(5):1633-1639. |
Rochkind S, Almog M, Meilin S, Nevo Z. Front Surg. 2021 Jan 15; 7:609638. |
On-site laser photobiomodulation treatment of crushed muscle due to prolonged pressure in rats. Almog M, Nissan M, Koifman I, Wollman Y, Rochkind S. |
Lasers Surg Med. 2021 Jun 8; Online ahead of print.
Huang Z, Kankowski S, Ertekin E, Almog M, Nevo Z, Rochkind S, Haastert-Talini K. Int J Mol Sci. 2021 Jun 18; 22(12):6554. |
Distribution Protective Effect of Photobiomodulation on Denervated and Crushed Muscle. Rochkind S and Almog A. Photomed Laser Surg, 2021 Jul 15; 39(7):435-436.
|
A rabbit model for peripheral nerve reconstruction studies avoiding automutilation Behaviour.
Sorkin JA, Rechany Z, Almog M, Dietzmeyer N, Shapira Y, Haastert-Talini K, Rochkind S. J Brachial Plex Peripher Nerve Inj. 2021; accepted.